
RATIONALE-306 evaluated outcomes with TEVIMBRA + chemotherapy vs placebo + chemotherapy in patients with 1L, unresectable, advanced or metastatic ESCC1-4
This global trial was a randomized, double-blind, placebo-controlled, phase 3 trial conducted in 16 countries at 162 sites across multiple continents: Asia, Europe, North America, and Oceania*
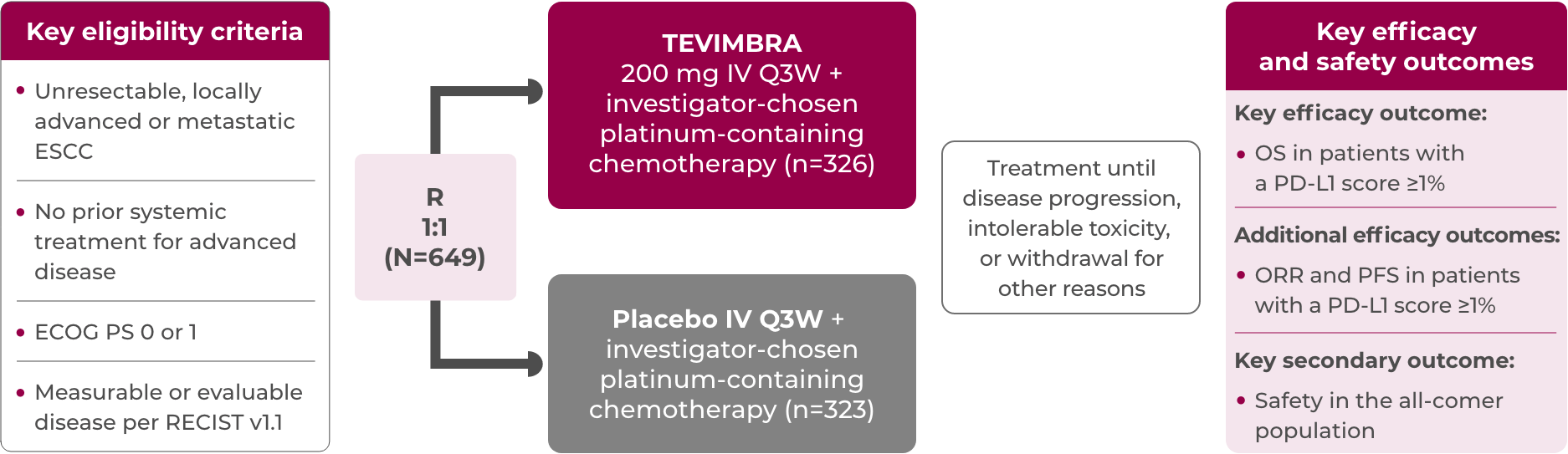
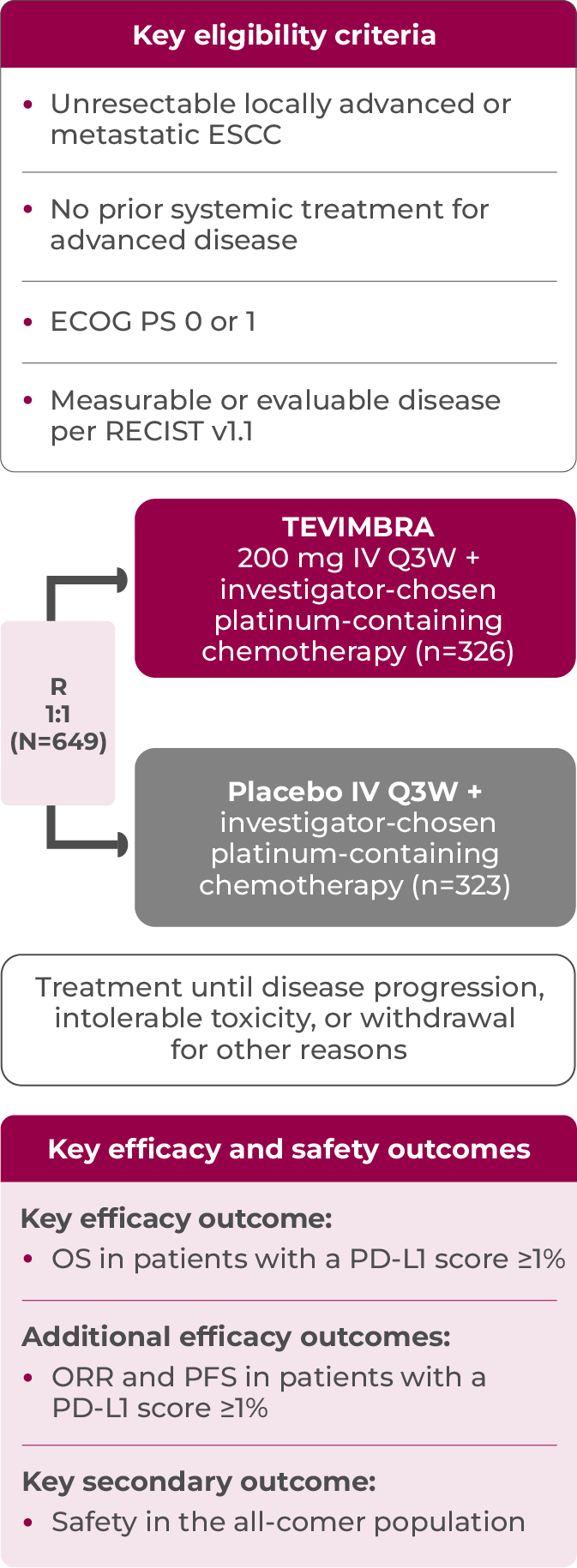
Key efficacy analysis in patients with a PD-L1 score ≥1%†: n=481
Safety analysis population: n=645
The patients of RATIONALE-306 were stratified by:
- Investigator-chosen chemotherapy (platinum + fluoropyrimidine vs platinum + paclitaxel)
- Region (Asia [excluding Japan] vs Japan vs rest of world)
- Previous definitive therapy (yes vs no)
TEVIMBRA was studied in multiple chemotherapy combinations—including oxaliplatin + 5-FU and CAPOX—allowing flexible dosing options for patients’ unique needs
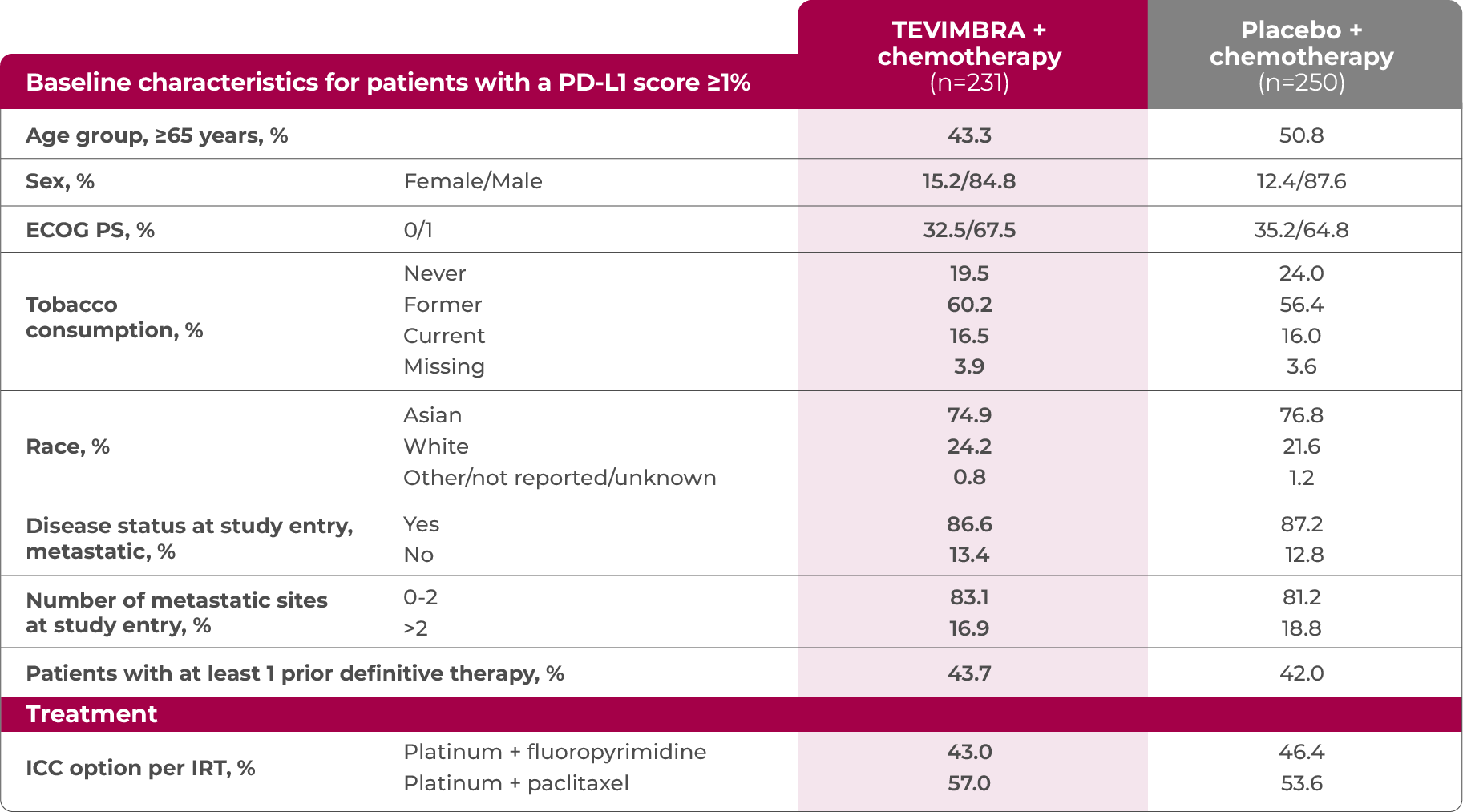
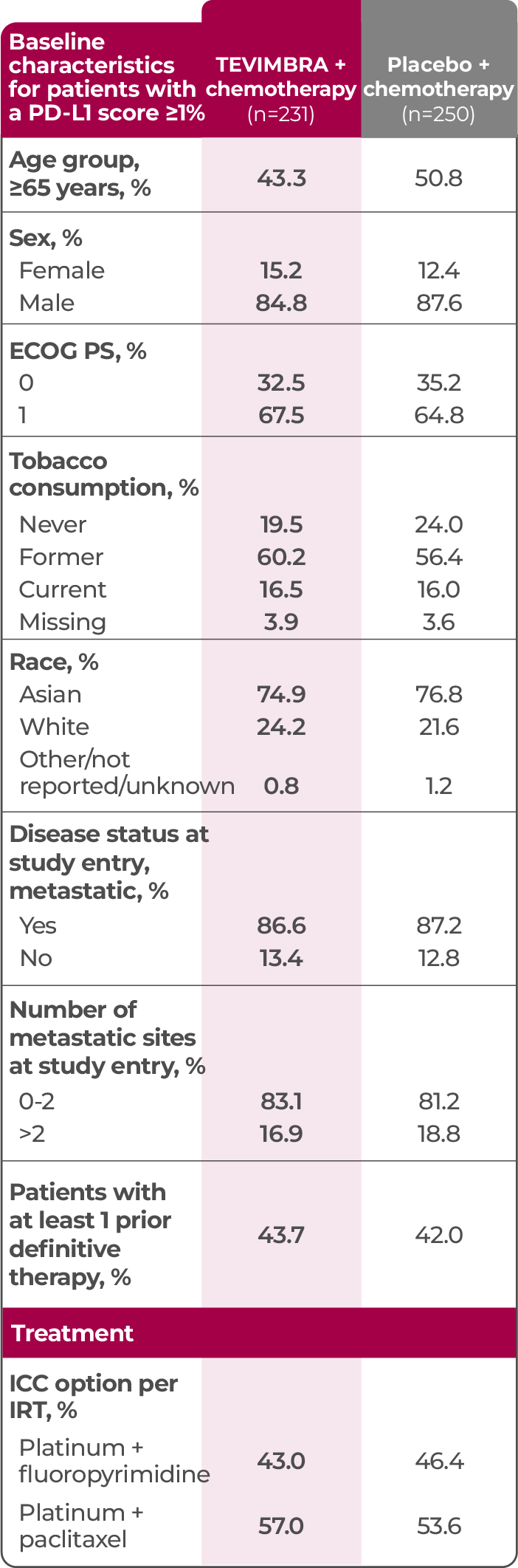
TEVIMBRA + chemotherapy was tested across a broad range of patients with a PD-L1 score ≥1%4
PD-L1 scores were measured in RATIONALE-306 using TAP, or tumor area positivity, which demonstrated substantial concordance with CPS, with ~90% overall agreement1,5
You may use your preferred PD-L1 testing method to identify patients in clinical practice
*Oceania included Australia.
†The key efficacy analysis was a retrospective subgroup analysis of RATIONALE-306 in patients with a PD-L1 score ≥1%. PD-L1–negative patients were excluded from this analysis population due to TEVIMBRA + chemotherapy being indicated only in patients whose tumors expressed PD-L1 (≥1). Therefore, the efficacy data shared in this presentation do not reflect the all-comer population.
References: 1. TEVIMBRA. Prescribing Information. BeOne Medicines USA, Inc.; 2025. 2. Xu J, Kato K, Raymond E, et al. Lancet Oncol. 2023;24(5):483-495. doi:10.1016/s1470-2045(23)00108-0 3. Xu J, Kato K, Raymond E, et al. Lancet Oncol. Supplementary appendix. 2023;24(5):483-495. doi:10.1016/s1470-2045(23) 00108-0 4. Xu J, Kato K, Hubner R, et al. Adv Ther. Published online March 13, 2025. doi:10.1007/s122325-025-03115-9 5. Moehler M, Yoon HH, Wagner DC, et al. Mod Pathol. 2025;38(9):100793. doi:10.1016/j.modpat.2025.100793
