
Proven to extend overall survival in patients with 1L GC/GEJC and PD-L1 scores ≥1%1
Key Efficacy Outcome: OS in PD-L1 ≥1%
15.0 months of median overall survival with TEVIMBRA + chemotherapy vs 12.8 months with placebo + chemotherapy
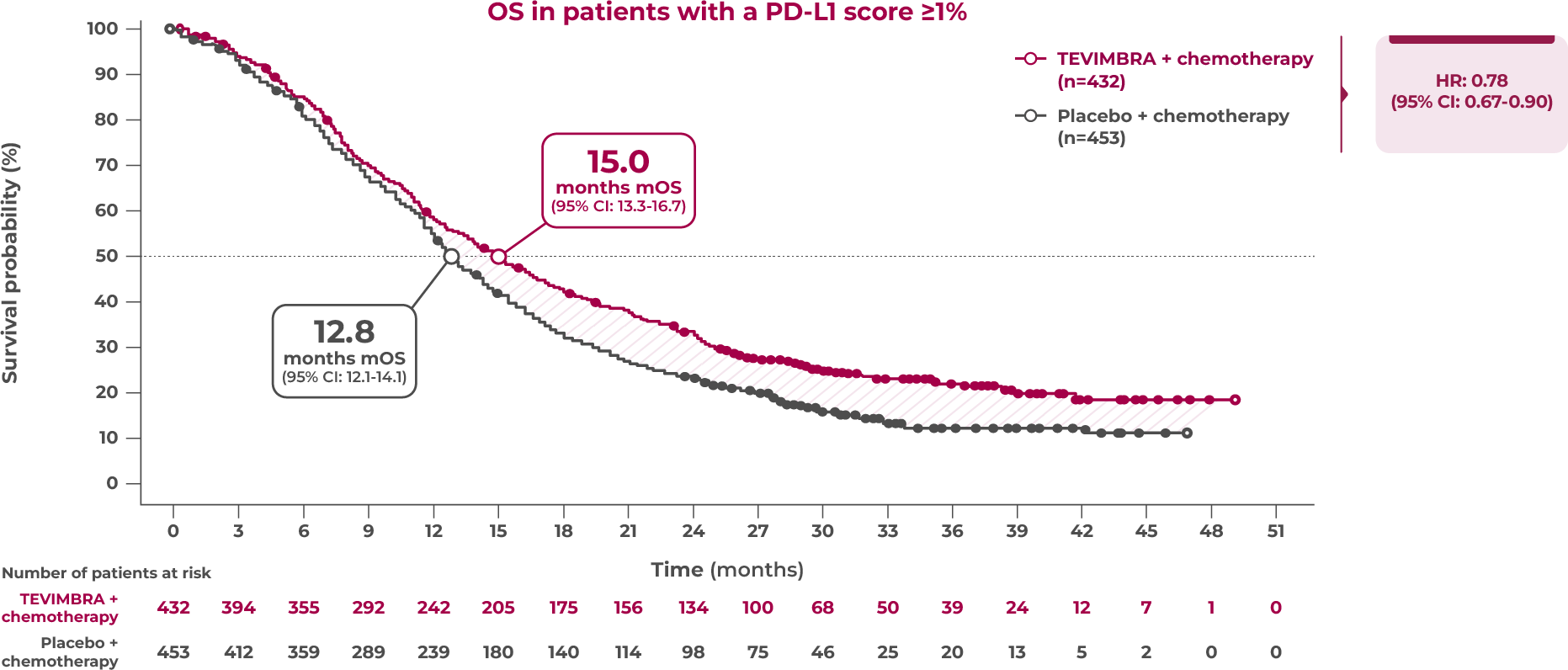
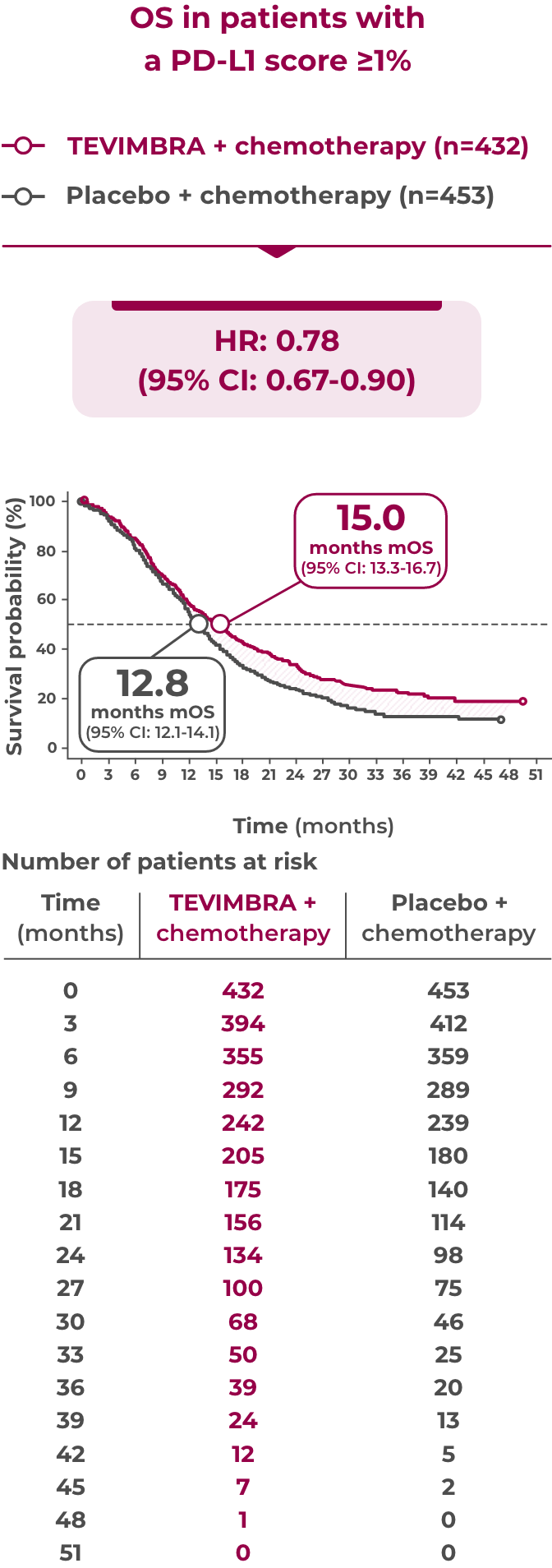
Limitation: Efficacy analysis was not powered for statistical comparison and is descriptive only. No definitive conclusions can be drawn.
3-Year Exploratory Follow-Up: OS in PD-L1 ≥1%
Overall survival sustained over 3 years4
21.3% of patients were still alive at 3 years with TEVIMBRA + chemotherapy (95% CI: 17.5-25.4) vs 13.1% with placebo + chemotherapy (95% CI: 10.1-16.5)
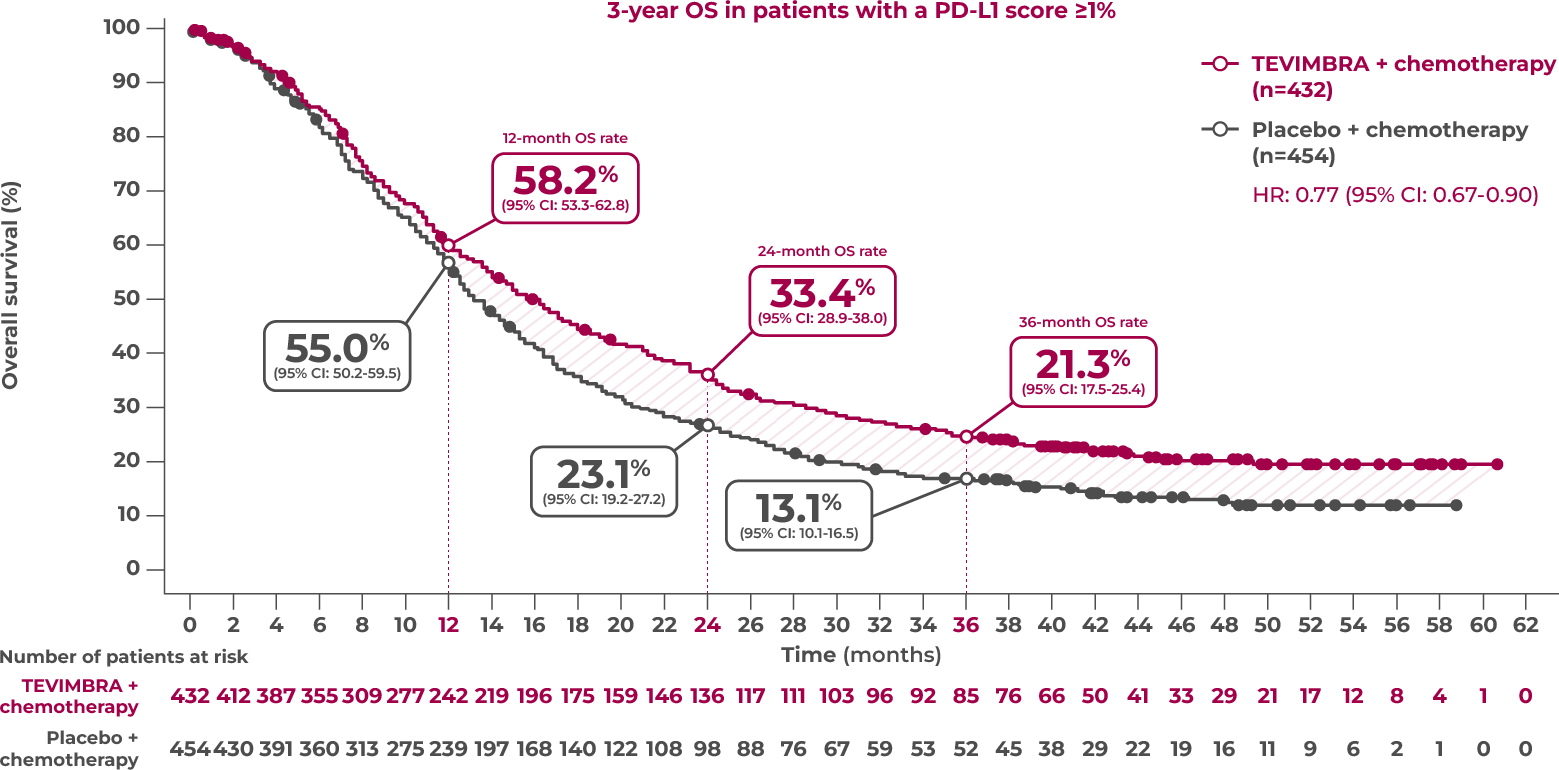
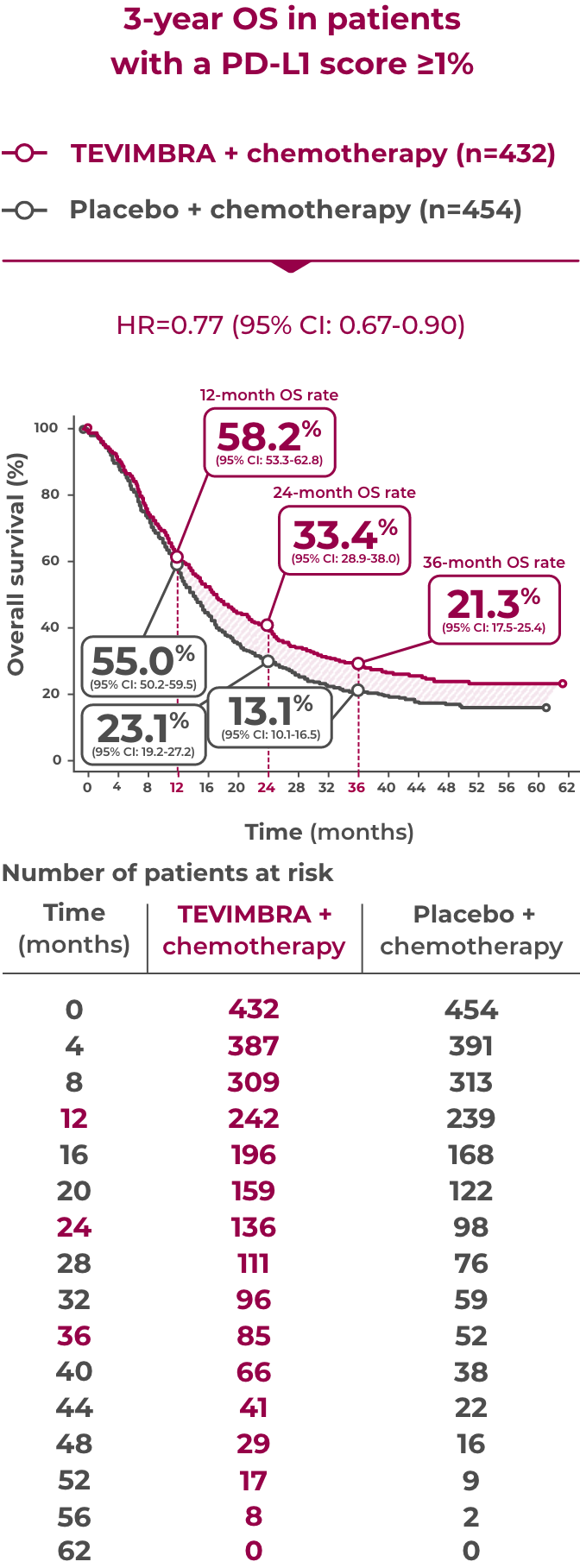
Limitation: The 3-year OS analysis was exploratory in nature and was not powered to show statistical significance. Landmark OS rates were estimated using the Kaplan-Meier method. No definitive conclusions can be drawn.
Subgroup Analyses: OS in PD-L1 ≥1%
A consistent trend in overall survival favoring TEVIMBRA + chemotherapy across multiple key subgroups4
TEVIMBRA + chemotherapy reported survival across common sites of metastases
- TEVIMBRA + chemotherapy is the first and only immunotherapy and chemotherapy treatment with positive survival data reported for patients with peritoneal metastases in a clinical trial using a PD-L1 score cutoff of ≥1%5-8

Analyses of OS*
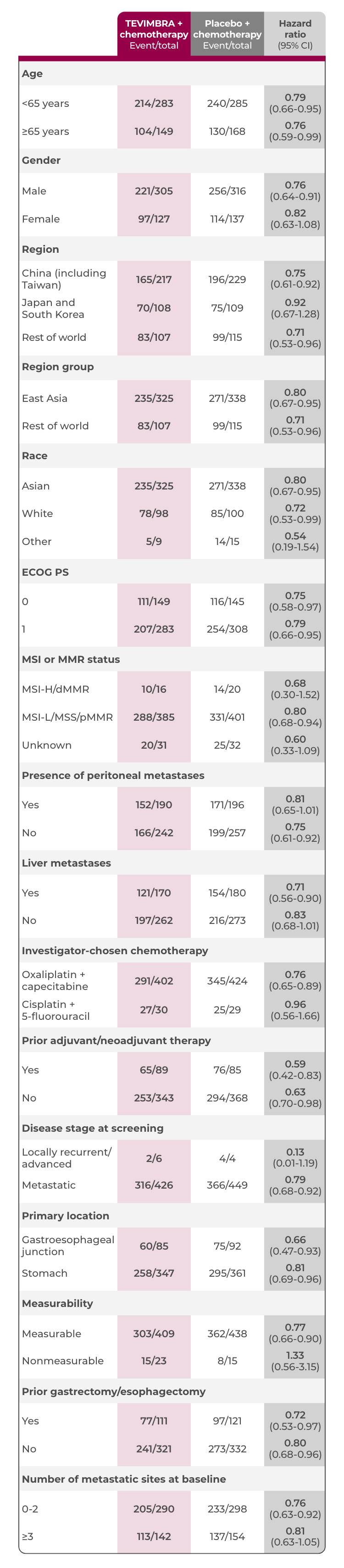
Limitation: Post-hoc subgroup analyses were not statistically powered and were descriptive only. No definitive conclusions can be drawn.
1L, first line; dMMR, deficient mismatch repair; ECOG, Eastern Cooperative Oncology Group; GC, gastric cancer; GEJ, gastroesophageal junction; GEJC, gastroesophageal junction cancer; HR, hazard ratio; MMR, mismatch repair; mOS, median overall survival; MSI, microsatellite instability; MSI-H, microsatellite instability high; MSI-L, microsatellite instability low; MSS, microsatellite stable; ORR, overall response rate; OS, overall survival; PD-L1, programmed death ligand 1; PFS, progression-free survival; pMMR, proficient mismatch repair; PS, performance status.
References: 1. TEVIMBRA. Prescribing Information. BeOne Medicines USA, Inc.; 2025. 2. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Esophageal and Esophagogastric Junction Cancers V.2.2026. © National Comprehensive Cancer Network, Inc. 2026. All rights reserved. Accessed February 25, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 3. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Gastric Cancer V.2.2026. © National Comprehensive Cancer Network, Inc. 2026. All rights reserved. Accessed February 25, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org. 4. Moehler M, Oh DY, Kato K, et al. Adv Ther. 2025;42(5):2248-2268. 5. Rha SY, Oh DY, Yañez P, et al; KEYNOTE-859 Investigators. Lancet Oncol. 2023;24(11):1181-1195. doi:10.1016/S1470-2045(23)00515-06 Published correction appears in Lancet Oncol. 2024;25(12):e626. doi:10.1016/S1470-2045(24)00650-8 6. Janjigian YY, Ajani JA, Moehler M, et al. J Clin Oncol. 2024;42(17):2012-2020. doi:10.1200/JCO.23.01601 7. Shitara K, Moehler M, Ajani J, et al. Poster presented at: American Society of Clinical Oncology Gastrointestinal Cancers Symposium; January 18-20, 2024; San Francisco, CA. Poster Bd E6. 8. Sonbol MB, Rha SY, Xu RH, et al. Abstract and poster presented at: ASCO Gastrointestinal Cancers Symposium; January 8-10, 2026. San Francisco, CA. Abstract 378, poster D9.
